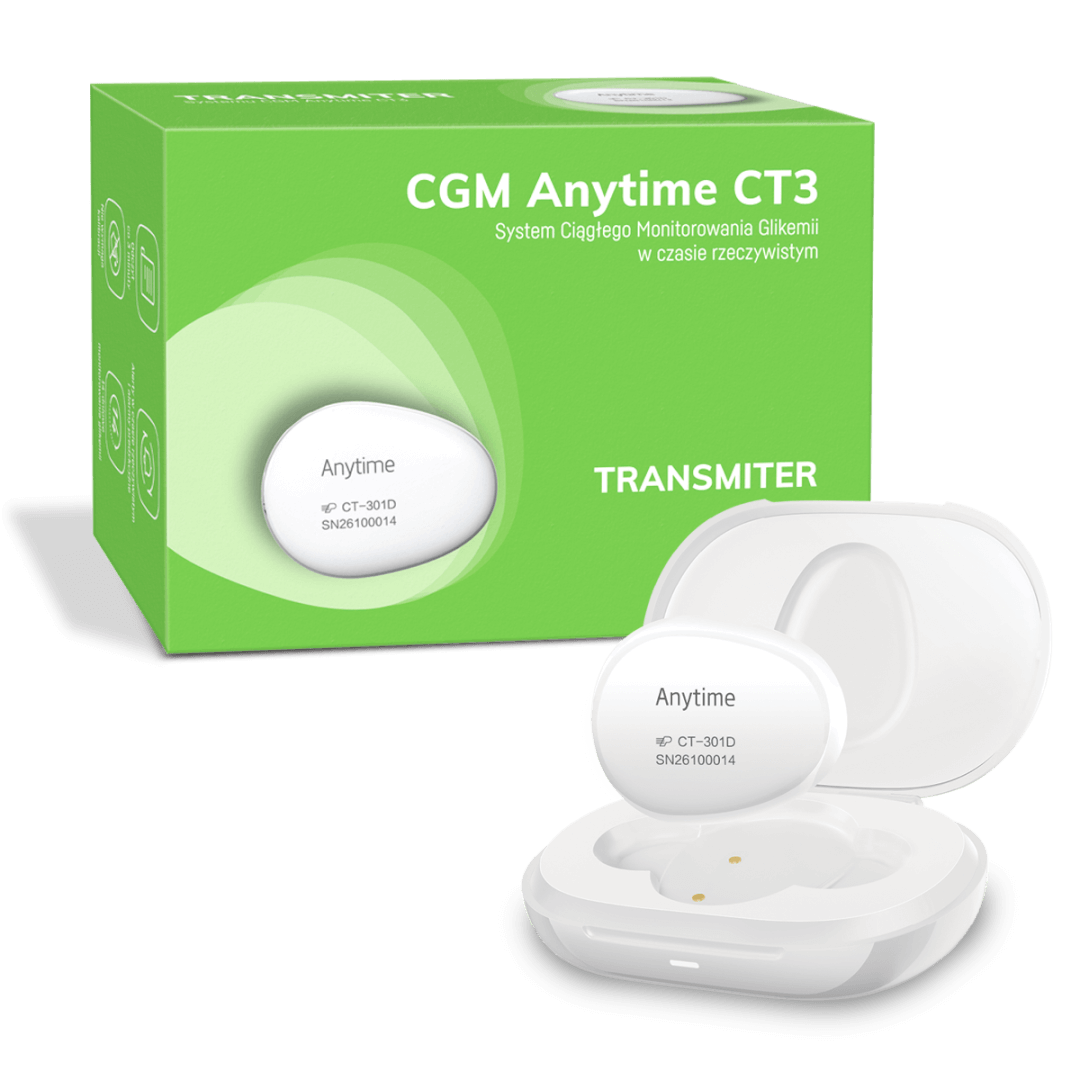
The Anytime CT3 continuous glucose monitoring (CGM) system – Transmiter
The Anytime CT3 continuous glucose monitoring (CGM) system enables real-time tracking of blood glucose levels 24 hours a day, anytime and anywhere.
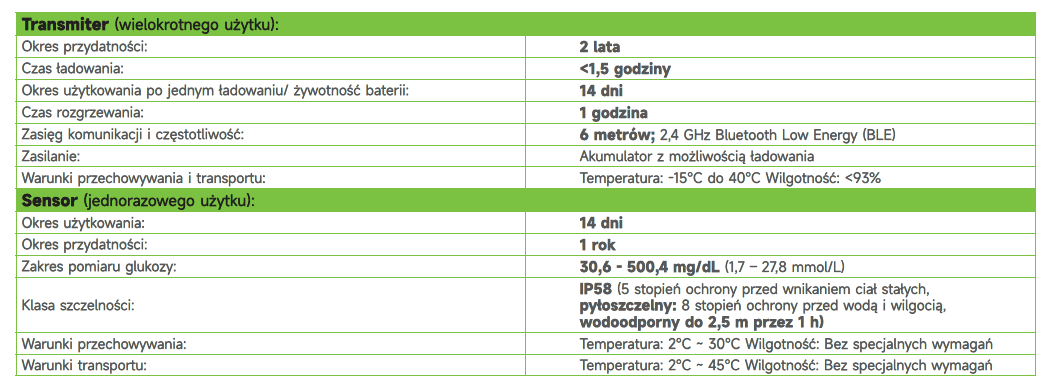
The transmitter is a component of the Anytime CT3 CGM system. It is reusable, with a service life of up to 2 years. The transmitter automatically sends glucose level data every 3 minutes to a free smartphone app.
ANYTIME CT3 CGM SYSTEM
- Can be used by patients aged 18 years and older, regardless of diabetes type and treatment regimen
- Enables insulin dosing based on CGM readings*
- No calibration required
- Allows selection of the application site: back of the upper arm, abdomen
- Features predictive Alerts and Alarms (20 minutes before a hypoglycemia event and 30 minutes before hyperglycemia)
- Wide glucose measurement range: 30.6–500.4 mg/dL (1.7–27.8 mmol/L)
- Dustproof and waterproof up to 2.5 meters for 1 hour (IP58 protection rating)
- Automatic interference correction for substances such as paracetamol and ascorbic acid
*Please note that insulin dosing, for any CGM system, should be based on treatment guidelines provided by the attending physician.
MEDICAL DEVICE – Continuous Glucose Monitoring System